Bioengineered Cornea

According to the WHO, corneal opacity accounts for over 4% of blindness worldwide, leading to more than 1.5–2 million cases each year

Globally, it is estimated that 4.9 million suffer from bilateral corneal blindness and 23 million from unilateral

Greater than 12.7 million people are waiting for a corneal transplant globally

India has a huge burden of bilateral (1.2 million) and Unilateral (5 - 6 million) corneal blindness. Each year, the burden increases by 30K new cases of blindness.
More than half of the donated corneas do not meet standards for transplant.
In order to address various kinds of corneal diseases and defects, Pandorum has developed Bioengineered Cornea – a class of products powered by tissue regenerative Exosomes and tissue-mimetic biomaterials, in order to promote regeneration of healthy functional cornea
Bioengineered Lenticule

Kuragenx- the ‘Liquid’ Cornea
A rare yet severe condition of corneal blindness is corneal ulceration, where patients suffer from inflammation, fibrosis, stromal melting, and damage to the nerves. Such serious cases of corneal damage, like a subset known as Neurotrophic keratitis, have high risk of transplant rejection and are even considered untreatable.
We have developed an advanced regenerative therapeutic option, the Kuragenx, comprising exosomes specialized for promoting cornea regeneration, combined with biopolymeric solution. This can be applied as drops on the cornea that solidifies in less than 10 min using visible light. In this formulation, the biopolymer acts as sacrificial matrix that allows sustained release of exosomes which supresses the ulceration defining condition like inflammation, reverses fibrosis, regenerates nerves and restores stromal thickness.
Kuragenx is our flagship product that can reverse an ulcerative cornea to a healthy state in less than a year. It has been developed with our excellent clinical partners in the US and India. In the US we have Dr. Satish Nadig and Dr. Ramez Haddadin, Northwestern University-Feinberg School of Medicine, Chicago and in India Dr. Virender S. Sangwan, Dr. Shroff’s charity eye hospital, New Delhi as our clinical collaborators.
Our exciting work and progress in developing this transformative technology got a special place in an article in Fortune India
Animal studies – Application of Kuragenx
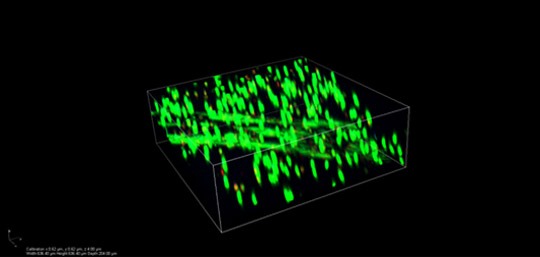
Uniform distribution and proliferation of cells inside Kuragenx